Tuesday, July 21, 2015
Neuropeptides and Stress
Select Neuropeptides can help moderate stress and related Alcohol seeking behaviors. The authors of this study reference use of our ppENK and proDYN Neuropeptides: J.R. Schank,B.S. Nelson,R. Damadzic,J.D. Tapocik,M. Yao,C.E. King,K.E. Rowe,K. Cheng,K.C. Rice,M. Heili. Neurokinin-1 receptor antagonism attenuates neuronal activity triggered by stress-induced reinstatement of alcohol seeking. doi:10.1016/j.neuropharm.2015.07.009.
Image: Mouse striatum stained with D2 cell marker Enkephalin (RA14124) in green and with neuronal marker NeuN in red courtesy of Dr Heike Rebholz of City College of New York.
Our Pain/Inflammation Antibodies are widely used and frequently referenced in studies of stress and the intersection between stress and pain.
Monday, June 22, 2015
Voices of our Customers
Our follow up processes include actively engaging users of our solutions for feedback and input. This includes Rose Ludescher, Manager of Customer Satisfaction, calling or sending e-mails to each user within 3 weeks of shipment. At the core is our product rating website. If users identify products related issues, we replace them or refund their purchases.
We want to hear from you...the good, the bad and the ugly. We welcome opportunities to fix issues.
We are always pleased when customers share data with us. Here's a recent example.
Image: Mouse striatum stained with D2 cell marker Enkephalin (RA14124) in green and with neuronal marker NeuN in red courtesy of Dr Heike Rebholz of City College of New York.
We are always delighted when we discover our solutions reference in Customer Publications. We try and post all of these to our website. We also post many here in recognition of our customers' research.
For all customers that share data or testimonials, we provide gift cards. It our way of saying thank you, because your feedback matters!
Wednesday, July 23, 2014
Pain Research Gene Expression Analysis
We can now add the GPNMB gene to the list of those anaylyzed: Lili Hou, Yanfeng Zhang, Yong Yang, Kai Xiang, Qindong Tan, Qulian Guo. Intrathecal siRNA Against GPNMB Attenuates Nociception in a Rat Model of Neuropathic Pain. Journal of Molecular Neuroscience. July 2014...Ten micrograms of siRNA1- GPNMB dissolved in 30 μl i-Fect transfection reagent (Neuromics, Edina, MN, USA) was administered intrathecally once daily for 7 days, starting from 1 day before CCI surgery...
Abstract: Neuropathic pain is characterized by hyperalgesia, allodynia, and spontaneous pain. Recent studies have shown that glycoprotein nonmetastatic melanoma B (GPNMB) plays a pivotal role in neuronal survival and neuroprotection. However, the role of GPNMB in neuropathic pain remains unknown. The aim of the present study was to assess the role of GPNMB in neuropathic pain. In cultured spinal cord neurons, we used two small interfering RNAs (siRNAs) targeting the complementary DNA (cDNA) sequence of rat GPNMB that had potent inhibitory effects on GPNMB, and siRNA1-GPNMB was selected for further in vivo study as it had the higher inhibitory effect. After sciatic nerve injury in rats, the endogenous level of GPNMB was increased in a time-dependent manner in the spinal cord. Furthermore, the intrathecal injection of siRNA1-GPNMB inhibited the expression of GPNMB and pro-inflammatory factors (TNF-α, IL-1β, and IL-6) and alleviated mechanical allodynia and thermal hyperalgesia in the chronic constriction injury (CCI) model of rats. Taken together, our findings suggest that siRNA against GPNMB can alleviate the chronic neuropathic pain caused by CCI, and this effect may be mediated by attenuated expression of TNF-α, IL-1β, and IL-6 in the spinal cord of CCI rats. Therefore, inhibition of GPNMB may provide a novel strategy for the treatment of neuropathic pain.
If you would like to learn how you can optimize your gene expression analysis studies, do not hesitate to e-mail: pshuster@neuromics.com or direct line: 612-801-1007.
Thursday, February 20, 2014
More MOR Publications
Our Mu Opioid Receptor Antibodies have set the standard for the study of Pain Mechanisms. We have posted >40 publications referencing use of these antibodies.
Here's several published in 2014:
Charlie H.T. Kwok, Ian M. Devonshire, Andrew J. Bennett, Gareth J. Hathway. Postnatal maturation of endogenous opioid systems within the periaqueductal grey and spinal dorsal horn of the rat. PAIN - January 2014 (Vol. 155, Issue 1, Pages 168-178, DOI: 10.1016/j.pain.2013.09.0220. ...rabbit anti-MOR (Neuromics, Edina, MN, USA; 1:1000 with tyramide signal amplification protocol)...
Images: Immunohistochemical expression of opioid peptides and receptors in the DH (spinal cord dorsal horn) during postnatal development. (A) POMC (pro-opiomelanocortin) immunoreactivity in the dorsal horn in postnatal day (P)10, P21, and adult rats. White arrows depict where cell staining was found. Interestingly, fibre staining was also observed in the superficial dorsal horn (lamina I) of adult rats, but not in the younger ages. (B) Since both cell and fibre staining were observed, staining intensity was used to quantify the immunoreactivity of POMC in the DH. Quantified staining intensity for POMC in the DH significantly decreased as the animals aged, with highest immunoreactivity found at P10. (C) Enkephalin immunoreactivity in the DH was restricted to the superficial laminae and only fibre staining was observed. (D) Quantified staining intensity for enkephalin illustrate an age-dependent increase in immunoreactivity, which was highest at adult P10.(C) Enkephalin immunoreactivity in the DH was restricted to the superficial laminae and only fibre staining was observed. (D) Quantified staining intensity for enkephalin illustrate an age-dependent increase in immunoreactivity, which was highest at adult. (E) MOR (μ-opioid receptor) immunoreactivity in the DH, cell staining was found throughout the superficial and deeper laminae in all ages. (F) Cell count of MOR staining in the DH, which showed a significant increase as the animals aged (∗∗P<0 .01="" adult="" i="" p21="" vs="">
J. Desroches, J.-F. Bouchard, L. Gendron, P. Beaulieu. Involvement of cannabinoid receptors in peripheral and spinal morphine analgesia ☆ Neuroscience, Volume 261, 7 March 2014, Pages 23–42. http://dx.doi.org/10.1016/j.neuroscience.2013.12.030. ...The floating sections were then incubated in 1% sodium borohydride in PBS for 30 min, rinsed twice with PBS, and incubated for 30 min at room temperature in a blocking solution containing 3% normal goat serum (NGS) and 0.3% Triton X-100 in PBS. The sections were then incubated overnight at 4 °C with the guinea pig anti-MOP primary antibody (cat# GP10106; Neuromics, Minneapolis, MN, USA) diluted 1:1000 in the blocking solution. The floating sections were then washed in PBS and incubated with a goat anti-guinea pig secondary antibody conjugated with Alexa Fluor 488 (Molecular Probes, Invitrogen, Carlsbad, CA, USA) at a concentration of 1:1000 in PBS for 2 h at room temperature...
We will continue posting customer data and publications that give new insights into the mechanisms of pain.
Saturday, October 12, 2013
P2X3 Receptor and Inflammatory Nociception
Endogenous ATP via activation of P2X3 Receptors contributes to inflammatory nociception in different models, including the formalin injected in subcutaneous tissue of the rat's hind paw. In this study, researchers evaluated whether TRPA1, 5-HT3 and 5-HT1A receptors, whose activation is essential to formalin-induced inflammatory nociception, are involved in the nociception induced by activation of P2X3 receptors on subcutaneous tissue of the rat's hind paw: Suzy Krimon, Dionéia Araldi, Filipe César do Prado, Cláudia Herrera Tambeli, Maria Cláudia G. Oliveira-Fusaro, Carlos Amílcar Parada. P2X3 receptors induced inflammatory nociception modulated by TRPA1, 5-HT3 and 5-HT1A receptors. Pharmacology Biochemistry and Behavior, Available online 8 October 2013. http://dx.doi.org/10.1016/j.pbb.2013.09.017. Our widely used and frequently published P2X3 R Antibody places a central role in measuring the expression of the protein...containing 5% non-fat dry milk at room temperature, followed by incubation with P2X3 rabbit polyclonal antibody (1:500; Neuromics) overnight at 4 °C, rinsed six times with TBST, and then incubated for 40 minutes in goat anti-rabbit IgG peroxidase...
Image: Neuromics' P2X3 R WB Example: Sequence‐specific siRNA‐mediated repression of P2X3. (A) P2X3 mRNA inhibition by 200 nM siRNA duplexes. Twenty‐four hours after transfection of CHO‐rP2X3 cells, P2X3‐specific mRNA was measured with Q‐PCR and plotted as percentage of mRNA detected in the control treated with Oligofectamine alone. Sequences and modifications are shown in Figure 1B and Table 1. (B) P2X3 protein reduction by 200 nM siRNA‐8646/8647, but not by its mismatch analogue siRNA‐MM‐7558/7559 or the unrelated siRNA‐7126/7127. Twenty‐four hours after transfection, protein was extracted and analysed by western blotting. P2X3‐specific immunodetection reveals expression levels as shown below (an average value from two experiments). Time points as indicated at the top. Molecular weights of two glycosylated forms of P2X3 are shown on the left.
Conclusions: Nociceptive response intensity was measured by observing the rat's behavior and considering the number of times the animal reflexively raised its hind paw (flinches) in 60 min. Local subcutaneous administration of the selective TRPA1, 5-HT3 or 5-HT1A receptor antagonists HC 030031, tropisetron and WAY 100,135, respectively, prevented the nociceptive responses induced by the administration in the same site of the non-selective P2X3 receptor agonist αβmeATP. Administration of the selective P2X3 and P2X2/3 receptor antagonist A-317491 or pretreatment with oligonucleotides antisense against P2X3 receptor prevented the formalin-induced behavioral nociceptive responses during the first and second phases. Also, the co-administration of a subthreshold dose of αβmeATP with a subthreshold dose of formalin induced nociceptive behavior, which was prevented by local administration of tropisetron, HC 030031 or WAY 100, 135. These findings have demonstrated that the activation of P2X3 receptors induces inflammatory nociception modulated by TRPA1, 5-HT3 and 5-HT1A receptors. Also, they suggest that inflammatory nociception is modulated by the release of endogenous ATP and P2X3 receptor activation, which in turn, increases primary afferent nociceptor susceptibility to the action of inflammatory mediators via interaction with TRPA1, 5-HT3 and 5-HT1A receptors in the peripheral tissue.
I will continue to post pain and inflammation related studies that reference the use of our antibodies.
Thursday, January 10, 2013
Pain and the Interplay Between P2Y Receptors with P2X3
Alfredo Ribeiro-da-Silva and his team use our Purinergic Receptor Antibodies to the study role of their expression in Nociceptive and Neuropathic Pain. They referenced their use in 8 publications.
Based on their research, they suspect changes in P2X3 function under pathological conditions are more complex than simple up- or down-regulation of expression at the protein level. This would have profound implications for the dicovery of drugs that target P2X3 expression levels: Gary Mo, Jennifer C. Peleshok, Chang-Qing Cao, Alfredo Ribeiro-da-Silva and Philippe Séguéla. Control of P2X3 channel function by metabotropic P2Y2 UTP receptors in primary sensory neurons. Molecular Pharmacology Fast Forward. Published on December 18, 2012 as doi:10.1124/mol.112.082099.
Monday, December 10, 2012
Pain Research and Gene Expression Analysis
I would like to highlight an exciting new study referencing how knocking down Kv9.1 Potassium Channel Subunit in vivo mediates neuropathic pain: Christoforos Tsantoulas, Lan Zhu, Yasin Shaifta, John Grist, Jeremy P. T. Ward, Ramin Raouf, Gregory J. Michael, and Stephen B. McMahon. Sensory Neuron Downregulation of the Kv9.1 Potassium Channel Subunit Mediates Neuropathic Pain following Nerve Injury. The Journal of Neuroscience, 28 November 2012, 32(48): 17502-17513; doi: 10.1523/JNEUROSCI.3561-12.2012.
Highlights: Here, we report that the potassium channel subunit Kv9.1 is expressed in myelinated sensory neurons, but is absent from small unmyelinated neurons. Kv9.1 expression was strongly and rapidly downregulated following axotomy, with a time course that matches the development of spontaneous activity and pain hypersensitivity in animal models. Interestingly, siRNA-mediated knock-down of Kv9.1 in naive rats led to neuropathic pain behaviors. Diminished Kv9.1 function also augmented myelinated sensory neuron excitability, manifested as spontaneous firing, hyper-responsiveness to stimulation, and persistent after-discharge. Intracellular recordings from ex vivo dorsal root ganglion preparations revealed that Kv9.1 knock-down was linked to lowered firing thresholds and increased firing rates under physiologically relevant conditions of extracellular potassium accumulation during prolonged activity. Similar neurophysiological changes were detected in animals subjected to traumatic nerve injury and provide an explanation for neuropathic pain symptoms, including poorly understood conditions such as hyperpathia and paresthesias. In summary, our results demonstrate that Kv9.1 dysfunction leads to spontaneous and evoked neuronal hyperexcitability in myelinated fibers, coupled with development of neuropathic pain behaviors.

Kv9.1 knock-down triggers ectopic activity and a form of peripheral wind-up in response to stimulation. A, Schematic illustrating the positions of stimulating and recording electrodes. B, Example recordings from centrally disconnected L4/L5 strands demonstrating SA in Kv9.1 siRNA-treated or nerve-injured rats, but not in control (scrambled siRNA) animals. C, Frequency-dependent SEA (denoted by double arrowheads) in Kv9.1 siRNA-treated (middle) and injured (right), but not control (left) animals. This activity is not locked in time and can be seen in between stimulation events (vertical arrows on top of 5 Hz stimulation traces, only first 5 shown). Also note the prolonged after-discharge (AD) observed in siRNA-treated and injured animals. D, Percentage of units showing SA and SEA in control (n = 269), Kv9.1 siRNA-treated (n = 369) and injured (n = 176) animals (*p < 0.05, **p < 0.01, ***p < 0.001 vs control, χ2 test). E, Firing rate of SEA units at different stimulation frequencies (mean ± SEM; control, n = 4; siRNA, n = 22; injured, n = 17; *p < 0.05 vs control, two-way ANOVA with Tukey's). F, Quantification of AD rate per SEA unit (mean ± SEM; *p < 0.05 vs control, Mann–Whitney test).
Results propose that Kv9.1 downregulation after nerve injury may be the molecular switch controlling myelinated sensory neuron hyperexcitability. Intriguingly, a recent wide-genome association screen in humans identified a Kv9.1 polymorphism associated with susceptibility to develop chronic neuropathic pain after back surgery or leg amputation (Costigan et al., 2010), suggesting that the mechanisms described in our studies will be of direct clinical relevance to human pain. Future efforts to elucidate the precise pathways involved, combined with approaches aiming to compensate loss of Kv9.1 function, may create novel therapeutic opportunities for neuropathic pain management.
Sunday, October 21, 2012
P2X3 Receptor and CGRP Antibodies Immunostaining
Here, use of our rabbit anti-CGRP and guinea pig anti-P2X3 is referenced. Please note the high titer of these antibodies (dilution is 1:25,000): Abeer W Saeed, Alfredo Ribeiro-da-Silva. Non-peptidergic primary afferents are presynaptic to neurokinin-1 receptor immunoreactive lamina I projection neurons in rat spinal cord. Molecular Pain 2012, 8:64 doi:10.1186/1744-8069-8-64.
Images: CGRP, IB4 and P2X3 staining in transverse spinal cord sections. A and B show low magnification confocal images of CGRP-IR and IB4 positive (A) or P2X3-IR (B) fibers. C and D represent high magnification confocal images from the middle third of the lateromedial extent of the superficial dorsal horn. In C, note that there is limited co-localization of IB4 and CGRP (in yellow). Arrowheads show axonal varicosities (boutons) from nonpeptidergic fibers in lamina I, which do not co-localize CGRP immunoreactivity. The framed regions in A and B indicate the approximate regions from where C and D, respectively, were obtained (the latter originate from other sections). CGRP (in green); IB4 (in red); P2X3 (in red). Scale bar (A, B) = 200 μm; scale bar (C, D) = 20 μm
Tissue processing: The injection site at the level of the parabrachial nucleus was examined by cutting serial, 100 μm-thick coronal sections of the relevant brain region. The dorsal aspect of the L4-L5 spinal cord segment was cut into serial, 50 μm-thick horizontal sections (n = 10), 50 μm-thick parasagittal sections (n = 4) or 50 μm-thick transverse sections (n = 4). All sections were cut using a freezing sledge microtome (Leica, Richmond Hill, Ontario) and collected as freefloating in phosphate-buffered saline (PBS) with 0.2% Triton-X 100 (PBS + T). To block unspecific staining, all spinal cord sections were incubated, for one hour, in 10% normal donkey serum (NDS) (Jackson, West Grove, PA) in PBS + T at room temperature. Subsequently, the sections were placed in primary antibodies (or conjugated lectin IB4 - see below) for 48 hours at 4 °C. We used a mixture of 2 or 4 primary antibodies (each raised in a different species), or IB4, in PBS + T containing 5% NDS. Next, the sections were washed in PBS + T and then incubated in species-specific secondary antibodies that were raised in donkey and conjugated to either AlexaFluor 488, AlexaFluor 405, Rhodamine RedX or biotin. The sections were incubated in 3 different cocktails: #1) rabbit anti-CGRP at a 1:200 dilution (Sigma, St Louis, MO) and lectin IB4 conjugated to AlexaFluor 568 at a 1:200 dilution (Molecular Probes); #2) rabbit anti-CGRP and guinea pig anti-P2X3 at a 1:25,000 dilution (Neuromics, Edina, MN); #3) goat anti-CTb at a 1:5000 dilution (List Biological), rabbit anti-NK-1r at a 1:10000 dilution (Sigma, St Louis, MO), guinea pig anti-CGRP at a 1:8000 dilution (Peninsula, San Carlos, CA) and lectin IB4 conjugated to AlexaFluor 647 at a 1:200 dilution (Molecular Probes). All the sections were washed with PBS + T and then (for #1) incubated for 2 hours at room temperature with donkey anti-rabbit AlexaFluor 488; (for #2) incubated for 90 minutes in a biotin conjugated donkey anti-guinea pig IgG (Jackson Immunoresearch, West Grove, PA, 1:200). Further signal amplification was achieved by treating the sections with 1 hour incubation in an avidin-biotin (A + B) complex (Vectastain Elite ABC kit, Vector Laboratories) followed by tyramide (Perkin-Elmer, Norwalk, CT, 1:75) for 7 minutes. Sections were then incubated in streptavidin conjugated to AlexaFluor 568 (Molecular Probes, Eugene, OR, 1:200) and donkey anti-rabbit AlexaFluor 488; or (for #3) incubated for 2 hours at room temperature with secondary antibodies: donkey anti-goat Rhodamine Red X, donkey anti-rabbit AlexaFluor 488, and donkey anti-guinea pig AlexaFluor 405. Finally, sections were washed with PBS, mounted on gelatin-subbed slides and coverslipped with an anti-fading mounting medium (Aqua Polymount; Polysciences, Warrington, PA). Slides were stored at −4 °C pending further processing.
I will continue to publish outstanding customer data/images using our natibodies/markers.
Wednesday, September 12, 2012
ASIC3 and Osteoarthritis
Neuromics' foundation is built on providing reagents for pain researchers. I have posted the twists and turns via key publications and related data. Here's yet another success story with one of our Pain and Inflammation Research Antibodies.
Acid sensing ion channels (ASICs) are sodium-selective ion channels activated by low extracellular pH, and belong to the degenerin/epithelial Na+ channel superfamily. ASIC3 is the most sensitive to such a pH change [2,3], abundantly expressed in dorsal root ganglia (DRG) [4], and strongly correlated with pain. Here researchers show the role of ASIC3 in osteoarthritis: Masashi Izumi, Masahiko Ikeuchi, Qinghui Ji, Toshikazu Tani. Local ASIC3 modulates pain and disease progression in a rat model of osteoarthritis. Journal of Biomedical Science 2012, 19:77 doi:10.1186/1423-0127-19-77.
Highlights: OA was induced via intra-articular mono-iodoacetate (MIA) injection, and pain related behaviors were evaluated including weight bearing measured with an incapacitance tester and paw withdrawal threshold in a von Frey hair test. OA rats showed not only weight-bearing pain but also mechanical hyperalgesia outside the knee joint (secondary hyperalgesia). ASIC3 expression in knee joint afferents was significantly upregulated approximately twofold at Day 14. Continuous intra-articular injections of APETx2 inhibited weight distribution asymmetry and secondary hyperalgesia by attenuating ASIC3 upregulation in knee joint afferents. Histology of ipsilateral knee joint showed APETx2 worked chondroprotectively if administered in the early, but not late phase.
Images: Fast Blue labeling and immunohistochemistry staining for ASIC3 : (a-b) Naïve- model, (c-d) OA-model, (e-f) APETx2 administration to OA-model in early phase. Photos in each row are the same DRG. In (b),(d),(f), large arrows indicate Fast Blue labeled, ASIC3 immunoreactive (ASIC3-ir) DRG cells, while ASIC3-ir cells that were not labeled by Fast Blue are indicated by small arrowheads. More than 100 FB-labeled neurons were analyzed from 4 rats in each group. The percentage of ASIC3-ir knee joint afferents was 18 ± 3% (mean ± SD) in naïve models, 46 ± 4% in OA-models (p = 0.003), and 20 ± 5% in the early-phase APETx2 group (p = 0.006), respectively. Scale bar: 50 μm
Protocol: The [DRG] sections were blocked in 3% normal goat serum for 1 h, then incubated in primary antibody of ASIC3 (Neuromics; Edina, MN, GP 14015, 1:500) overnight in a humid chamber. The next day, the sections were incubated in the secondary antibody (Vector; Burlingame, CA, FI-7000, 1:500, FITC tagged) for 2 h. All antisera used were diluted in PBS containing 1% normal goat serum and 0.05% Triton X-100. Before, between, and after each incubation step, the sections were washed 3 times for 5 min in PBS. Finally, all sections were mounted with Vectashield (Vector, Burlingame, CA).
1. Waldmann R, Champigny G, Bassilana F, Heurteaux C, Lazdunski M: A proton-gated cation channel involved in acid-sensing. Nature 1997, 386:173–177. 2. Lingueglia E: Acid-sensing ion channels in sensory perception. J Biol Chem 2007, 282:17325–17329. note: see http://neuromics.net/weblog/post/tag/dr-eric-lingueglia/ for research using our siRNA transfectio reagent for ASIC3 gene expression analysis.
3. Wemmie JA, Price MP, Welsh MJ: Acid-sensing ion channels: advances, questions and therapeutic opportunities. Trends Neurosci 2006, 29:578–586.
4. Voilley N, de Weille J, Mamet J, Lazdunski M: Nonsteroid anti-inflammatory drugs inhibit both the activity and the inflammation-induced expression of acid-sensing ion channels in nociceptors. J Neurosci 2001, 21:8026–8033.
Sunday, May 20, 2012
Cannabinoid Type-1 Receptors and Chemotherapy Related Pain
The authors use our guinea pig TRPV1 antibody to measure cisplatin hyperalgesia vs treated and control mice.
Images: URB597 attenuated effects of cisplatin on protein expression in DRGs. A, TRPV1- and ATF-3-ir were detected by immunofluorescence in L3–L5 DRGs from mice treated with vehicle, cisplatin, or cisplatin plus URB597. Cisplatin (1 mg/kg of body weight, daily for 7 d, i.p.) increased the occurrence of TRPV1- and ATF3-ir in neurons. Co-injection of URB597 (0.3 mg/kg daily, i.p.) with cisplatin attenuated the effect cisplatin on protein-ir. Scale bars: 10 μm (for images within each antigen). B, Quantitative summary of the effect of treatments on TRPV1-ir in neurons. Data are expressed as the mean ± SEM. aSignificantly different from each other group (p < 0.05, one-way ANOVA with Student–Newman–Keuls test; n = 4 mice/treatment). C, Quantitative summary of the effect of treatments on ATF-ir in neurons. Data are expressed as the median and 25th and 75th percentile range. *Significantly different from vehicle control and cisplatin plus URB597 groups (n = 6 mice/treatment; p < 0.001, Kruskal–Wallis ANOVA on ranks test).
Related Reagents:
VR1 N-Terminus (TRPV1)
VR1 (TRPV1)-Goat
VR1 C-Terminus (TRPV1) - mouse specific
All TRP Antibodies
Pain and Inflammation Research Antibodies
Neurotransmission -Neurotransmission Research Antibody Categories
Thursday, May 17, 2012
Pinpointing Neuropathic Pain
In this study, the authors showed ablation of nonpeptidergic fibers in a chronic constriction injury model caused significant sympathetic and parasympathetic P2X3 fiber sprouting, and led to an exacerbated pain response. This was an unexpected finding, as it has been suggested that nonpeptidergic fibers play a major role in mechanical pain, and suggests that these fibers play a complex role in the development of neuropathic pain: Anna M.W. Taylora, Maria Osikowicza, Alfredo Ribeiro-da-Silva. Consequences of the ablation of nonpeptidergic afferents in an animal model of trigeminal neuropathic pain. PAIN. Volume 153, Issue 6, June 2012, Pages 1311–1319. doi.org/10.1016/j.pain.2012.03.023....Sections were then incubated for 48 hours at 4°C with a guinea pig polyclonal anti-P2X 3 antibody (1:25,000; Neuromics, Edina, MN, USA), diluted in PBS-T. Following primary antibody incubation, sections were treated with a biotin-conjugated...
Image: Example of Non-petidergic fibers expressing P2X3 receptor for ATP are present in lamina II of the contralateral dorsal horn (arrows). The P2X3-positive fibers are eliminated from the rat dorsal horn ipsilateral to the rhizotomy (arrow heads). Spinal cord segment C6. Scale bar 400 um. P2X3 antibody dilution 1:25,000. Biomédica vol.24 no.2.
Check out our related reagents categories:
- All Purinergic Receptors
- Pain and Inflammation Research Antibodies
- Neurotransmission Research Antibodies
- Primary Neurons and Astrocytes-Primary human, rat and mouse neurons and astrocytes
Monday, March 26, 2012
Pain Research-Pubs Update
Andrew C. Eschenroeder, Allison A. Vestal-Laborde, Emilse S. Sanchez, Susan E. Robinson, Carmen Sato-Bigbee. Oligodendrocyte responses to buprenorphine uncover novel and opposing roles of μ-opioid- and nociceptin/orphanin FQ receptors in cell development: Implications for drug addiction treatment during pregnancy. Glia Volume 60, Issue 1, pages 125–136, January 2012....The MOR and NOP receptor antibodies were from Neuromics (Edina, MN) and used for immunocytochemistry (1:100)and western blotting (1:500)...
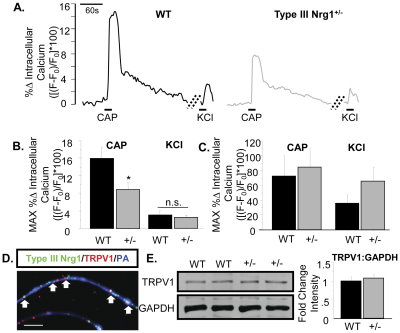
FiFigure. Sensory axons, but not soma, from Type III Nrg1+/− mice show reduced capsaicin responsiveness compared to axons from WT mice. (A) Representative traces of intracellular calcium along sensory axons in response to 1 µM capsaicin or 56 mM KCl. The change in intracellular calcium from baseline over time ([(F−F0)/F0]*100) is shown for WT (left) and Type III Nrg1+/− (right) axons. Hatched diagonal lines indicate where the time course was non-continuous. (B) Quantification of the maximum change in intracellular calcium in response to application of 1 µM capsaicin or 56 mM KCl by genotype. Averages of 5 animals per genotype were compared using a Student's t-test. Type III Nrg1+/− axons showed a significantly decreased response to capsaicin (p<0.05), but not to KCl, relative to WTs. Graph shows mean±SEM. (C) Type III Nrg1+/− sensory soma show normal response to capsaicin. Quantification of maximal change in fluorescence from baseline ([(F−F0)/F0]*100) in WT or Type III Nrg1+/− sensory neuron soma in response to 1 µM capsaicin or 56 mM KCl. Average responses from 4 WT and 4 Type III Nrg1+/− animals to application of capsaicin or KCl were compared by genotype using a Student's t-test. There was no statistically significant difference between genotypes. Graphs show mean±SEM. (D) Type III Nrg1 (green) and TRPV1 (red) are co-expressed along P21 WT cultured sensory neuron axons identified with a pan-axonal (PA) marker (blue). White arrows indicate examples where Type III Nrg1 and TRPV1 are in close proximity. Scale bar equals 10 µm. (E) P21 WT and Type III Nrg1+/− sensory neuron cultures have equivalent levels of total TRPV1 protein. Total TRPV1 protein measurement by immunoblot. The 95 kD TRPV1 band and the 35 kD GAPDH band are shown from a representative experiment comparing protein from P21 WT and Type III Nrg1+/− cultures. Quantification of fold change in intensity of TRPV1:GAPDH normalized to WT average. There was no statistically significant change in the ratio of TRPV1 to GAPDH between genotypes (WT, Type III Nrg1+/−, n = 3 animals). Genotype comparisons were made using a Student's t-test. Graph shows mean±SEM. doi:10.1371/journal.pone.0025108.g004
Related Links:
- Pain and Inflammation Research Antibodies
- Transfection Kits and Reagents-optimizing the delivery of siRNA and plasmids to neurons and the CNS.
- Primary Neurons and Astrocytes-Primary human, rat and mouse neurons and astrocytes
Wednesday, November 10, 2010
Our TRPV1 Antibodies Rock
VR1 N-Terminus (TRPV1)-Rabbit
T. Wu, L. Song, X. Shi, Z. Jiang, J. Santos-Sacchi and A.L. Nuttal. Effect of capsaicin on potassium conductance and electromotility of guinea pig outer hair cell. doi:10.1016/j.heares.2010.10.010
...anti-TRPV1 (rabbit polyclonal, RA10110, Neuromics, Edina, MN, USA) diluted to 1:500 with 1% BSA-PBS...antibody (TRPV-1) and its blocking peptide (104 M) (Neuromics, Edina, MN, USA)...
(TRPV1) - mouse specific
Julie A. Christianson, Klaus Bielefeldt, Sacha A. Malin and Brian M. Davis. Neonatal colon insult alters growth factor expression and TRPA1 responses in adult mice. Pain Volume 151, Issue 2, November 2010, Pages 540-549.
...primary antiserum to TRPV1 (1:4000; Neuromics, Minneapolis, MN; cat# RA14113)...
VR1 C-terminus (TRPV1)-Guinea Pig
N. Schuelert, C. Zhang, A.J. Mogg, L.M. Broad, D.L. Hepburn, E.S. Nisenbaum, M.P. Johnson and J.J. McDougal. Paradoxical effects of the cannabinoid CB2 receptor agonist GW405833 on rat osteoarthritic knee joint pain. Osteoarthritis and Cartilage Volume 18, Issue 11, November 2010, Pages 1536-1543.
...primary antiserum to TRPV1 (1:4000; Neuromics, Minneapolis, MN; cat# RA14113)...
Mariusz Mucha, Lezanne Ooi, John E. Linley, Pawel Mordaka, Carine Dalle, Brian Robertson, Nikita Gamper, and Ian C. Wood Transcriptional Control of KCNQ Channel Genes and the Regulation of Neuronal Excitability.
J. Neurosci., Oct 2010; 30: 13235 - 13245 ; doi:10.1523/JNEUROSCI.1981-10.2010
...1:1000 guinea pig anti-TRPV1 (Neuromics)...
Related Reagents:
All TRP Antibodies
Pain and Inflammation Research Antibodies
Neurotransmission -Neurotransmission Research Antibody Categories
Thursday, September 09, 2010
Potential Therapeutic Targets for Bone Cancer Pain-P2X Receptors
I would like to alert you to the latest reference:
Timothy K. Y. Kaan, Ping K. Yip, Sital Patel, Meirion Davies, Fabien Marchand, Debra A. Cockayne, Philip A. Nunn, Anthony H. Dickenson, Anthony P. D. W. Ford, Yu Zhong, Marzia Malcangio, and Stephen B. McMahon Systemic blockade of P2X3 and P2X2/3 receptors attenuates bone cancer pain behaviour in rats. Brain, September 2010; 133: 2549 - 2564.
......Slides were then incubated with rabbit anti-P2X3 (1:2000, Neuromics) and sheep anti-calcitonin gene-related peptide (1:1000, Biomol...anti-beta-III-tubulin (1:4000, Promega) and guinea pig anti-P2X3 (1:100, Neuromics). The next day, after three washes with phosphate-buffered......
Summary: Pain remains an area of considerable unmet clinical need, and this is particularly true of pain associated with bone metastases, in part because existing analgesic drugs show only limited efficacy in many patients and in part because of the adverse side effects associated with these agents. An important issue is that the nature and roles of the algogens produced in bone that drive pain-signalling systems remain unknown. Here, we tested the hypothesis that adenosine triphosphate is one such key mediator through actions on P2X3 and P2X2/3 receptors, which are expressed selectively on primary afferent nocioceptors, including those innervating the bone. Using a well-established rat model of bone cancer pain, AF-353, a recently described potent and selective P2X3 and P2X2/3 receptor antagonist, was administered orally to rats and found to produce highly significant prevention and reversal of bone cancer pain behaviour. This attenuation occurred without apparent modification of the disease, since bone destruction induced by rat MRMT-1 carcinoma cells was not significantly altered by AF-353. Using in vivo electrophysiology, evidence for a central site of action was provided by dose-dependent reductions in electrical, mechanical and thermal stimuli-evoked dorsal horn neuronal hyperexcitability following direct AF-353 administration onto the spinal cord of bone cancer animals. A peripheral site of action was also suggested by studies on the extracellular release of adenosine triphosphate from MRMT-1 carcinoma cells. Moreover, elevated phosphorylated-extracellular signal-regulated kinase expression in dorsal root ganglion neurons, induced by co-cultured MRMT-1 carcinoma cells, was significantly reduced in the presence of AF-353. These data suggest that blockade of P2X3 and P2X2/3 receptors on both the peripheral and central terminals of nocioceptors contributes to analgesic efficacy in a model of bone cancer pain. Thus, systemic P2X3 and P2X2/3 receptor antagonists with central nervous system penetration may offer a promising therapeutic tool in treating bone cancer pain.
Related Reagents:
All Purinergic Receptors
Neurotransmission Research Antibodies
Thursday, July 29, 2010
Let-7 microRNAs and Nociceptive Pain
We want to recognize Dr. Zaijie Jim Wang and his team for being the first to use our Mu Opioid Receptor for studying the potential role of microRNAs in Nociception.
Ying He, Cheng Yang, Chelsea M. Kirkmire, and Zaijie Jim Wang. Regulation of Opioid Tolerance by let-7 Family MicroRNA Targeting the µ Opioid Receptor. The Journal of Neuroscience, July 28, 2010, 30(30):10251-10258; doi:10.1523/JNEUROSCI.2419-10.2010
Abstract: MicroRNA has emerged as a critical regulator of neuronal functions. This study aimed to test whether let-7 microRNAs can regulate the µ opioid receptor (MOR) and opioid tolerance. Employing bioinformatics, we identified a let-7 binding site in the 3'-untranslated region (UTR) of MOR mRNA, which was experimentally confirmed as a direct target of let-7. The repressive regulation of MOR by let-7 was revealed using a LNA-let-7 inhibitor to knockdown let-7 in SH-SY5Y cells. Conversely, morphine significantly upregulated let-7 expression in SH-SY5Y cells and in a mouse model of opioid tolerance. The LNA-let-7 inhibitor decreased brain let-7 levels and partially attenuated opioid antinociceptive tolerance in mice. Although chronic morphine treatment did not change overall MOR transcript, polysome-associated mRNA declined in a let-7-dependent manner. let-7 was identified as a mediator translocating and sequestering MOR mRNA to P-bodies, leading to translation repression. These results suggest that let-7 plays an integral role in opioid tolerance.
- Western blot analysis. Western blot analysis was performed as previously described (Tang et al., 2006) using the anti-µ opioid receptor antibody (1:1000; Neuromics). The expression of β-actin was similarly determined from the same blots using a monoclonal antibody (1:10,000; Sigma).
- For immunofluorescence analysis, the antibody for hDcp1a (Santa Cruz Biotechnology) and MOR were used at 1:500 and 1:5000 dilutions, respectively. Secondary anti-goat and anti-mouse antibodies labeled with Alexa 488 and Alexa 594 fluorochromes (Invitrogen), respectively, were used at 1:500 dilutions.
All Opioid Receptor Antibodies
Pain and Inflammation
Neurotransmission Research Antibodies-GPCRs, Ligand Gated Ion Channels,
Biogenic Amines and more
i-Fect Transfection Kit -gene silencing of DOR,
NaV1.8 tetrodotoxin-resistant sodium channel, NTS2 and more in-vitro and in vivo
Primary Neurons and Astrocytes-Primary human,
rat and mouse neurons and astrocytes
Tuesday, July 06, 2010
TRPV1 & P2X3-Daily Double
Saturday, October 17, 2009
More Success with TRPV1
The antibody was used for both Western Blot and Immunohistochemistry. Here's a recent publication of their results:
Vanessa Bockhart, Cristina Elena Constantin, Annett Häussler, Nina Wijnvoord, Maike Kanngiesser, Thekla Myrczek, Geethanjali Pickert,Laura Popp, Jürgen-Markus Sobotzik, Manolis Pasparakis, Rohini Kuner, Gerd Geisslinger, Christian Schultz, Michaela Kress, and Irmgard Tegeder. Inhibitor B Kinase β Deficiency in Primary Nociceptive Neurons Increases TRP Channel Sensitivity. The Journal of Neuroscience, October 14, 2009, 29(41):12919-12929; doi:10.1523/JNEUROSCI.1496-09.2009
Related Reagents:
All TRPV (Vanilloid); TRPM; TRPA and TRPC Antibodies
Pain and Inflammation Research Antibodies
Monday, September 28, 2009
Prodynorphin at Work

Image: Staining of adult rat spinal cord.
The tissue is perfusion-fixed (4% freshly prepared formaldehyde) adult rat spinal cord, reacted overnight with the PPD at 1:1000 and then o/n in Alexa488 secondary (raised in donkey, Invitrogen, 1:500).
The confocal image stack was taken through a 60x oil lens (Bio-Rad Radiance confocal) - pixel size is 0.196 micrometre and this is a projection of 10 confocal optical sections at 0.5 micrometre z-spacing.
Customer Publications
Related Reagents:
proDynorphin (guinea pig)
Opioid Receptors
Pain and Inflammation Antibodies
Thursday, September 10, 2009
TRPV1 Antibodies in Action

Images: The CCR2 chemokine receptor colocalized with IB4 and TRPV1, markers of nociceptive neurons, after injury. A) Many lumbar DRG neurons in vehicle-treated rat sensory neurons were positive for IB4, a neuronal phenotype that distinguishes some C-fiber nociceptors (red cells), however there was no expression of the CCR2 protein. B) After perineural gp120/hCD4 treatment, CCR2 protein expression (green cells) was upregulated, and co-localized with IB4. C) Both gp120/hCD4 and ddC treatment resulted in an upregulation of CCR2 expression (green cells) in many small and medium diameter neurons. Again, CCR2 co-localized in a number of IB4 positive cells. D) The TRPV1 channel is present on many nociceptive neurons and is involved in the neuropathic pain mechanism. Under normal conditions, TRPV1 was expressed in neurons (red cells). E) After gp120/hCD4 treatment, CCR2 expression is upregulated (green cells) and colocalized with TRPV1. F) After the combination of gp120/hCD4 and ddC treatments, again CCR2 was upregulated to a similar degree as gp120/hCD4 treatment alone and exhibited some colocalizations with TRPV1.
Scale Bar: 100um.
We would also like to share recent publications referencing our TRPVs:
Hao Sun, De-Pei Li, Shao-Rui Chen, Walter Hittelman, and Hui-Lin PanSensing of Blood Pressure Increase by Transient Receptor Potential Vanilloid 1 Receptors on BaroreceptorsJ. Pharmacol. Exp. Ther., Sep 2009; doi:10.1124/jpet.109.160473 ......guinea pig anti-TRPV1, dilution 1:1000, Neuromics, Minneapolis, MN) and secondary antibody...guinea pig anti-VR1 C-terminus (TRPV1), dilution 1:1000,Neuromics; and rabbit anti-NF200, dilution 1:100...guinea pig anti-TRPV1, dilution 1:1000, Neuromics) for 2 hr at room temperature and
overnight......Kenjiro Matsumoto, Emi Kurosawa, Hiroyuki Terui, Takuji Hosoya, Kimihito Tashima, Toshihiko Murayama, John V. Priestley, and Syunji HorieLocalization of TRPV1 and contractile effect of capsaicin in mouse large intestine: high abundance and sensitivity in rectum and distal colonAm J Physiol Gastrointest Liver Physiol, Aug 2009; 297: G348 - G360. ......TRPV1 antibody (mouse TRPV1 C-terminus; Neuromics, Minneapolis, MN) were 1:60,000 for rectum...different anti-TRPV1 antibodies (1:60,000, Neuromics, and rat TRPV1 COOH-terminus, 1:1,000...experiments, the antibody (1:60,000; Neuromics) was preincubated with 10 uM of the corresponding......
Featured Reagents:
VR1 N-Terminus (TRPV1)
VR1 C-terminus (TRPV1)
VR1 C-Terminus (TRPV1) - mouse specific
Related Reagents
VR1 (TRPV1)-Goat
VR like-3 (TRPV3)
All TRPV (Vanilloid); TRPM; TRPA and TRPCs
Pain and Inflammation Antibodies
Monday, July 27, 2009
TRPA1 in Craniofacial Muscle Pain
Jin Y. Ro, Jong-Seok Lee and Youping Zhang. Activation of TRPV1 and TRPA1 leads to muscle nociception and mechanical hyperalgesia. doi:10.1016/j.pain.2009.04.021.
... TRPA1 (1:1500; rat polyclonal, Neuromics)...
Featured Reagents
TRPA1-Cat#: RA14135
Related Reagents:
TRPA1 for WB
VR1 N-Terminus (TRPV1)
VR1 C-terminus (TRPV1)
VR1 C-Terminus (TRPV1) - mouse specific
VR1 (TRPV1)-Goat
VR like-3 (TRPV3)
All TRPV (Vanilloid); TRPM; TRPA and TRPCs
Pain and Inflammation Antibodies















