Wednesday, December 28, 2016
Dopamine and Morphine Tolerance
Our Mu Opioid Receptor antibody is used to show that blocking dopamine decreases morphine tolerance: Wen-Ling Dai, Feng Xiong, Bing Yan, Zheng-Yu Cao, Wen-Tao Liu, Ji-Hua Liu1, Bo-Yang Yu. Blockade of neuronal dopamine D2 receptor attenuates morphine tolerance in mice spinal cord. Scientific Reports 6, Article number: 38746 (2016). doi:10.1038/srep38746.
Images: (A) Double immunofluorescence staining showed that MOR (green) and D2DR (red) were co-localized in the mice spinal cord (20X magnification). Chronic morphine treatment increased the co-localization of MOR and D2DR in the spinal cord, and D2DR antagonist sulpiride (8 μg/10 μl, i.t.) reduced the increased co-expression of D2DR with MOR (n = 4). (B) Co-IP experiments showed that D2DR could interact with MOR, and the MOR/D2DR interactions were increased in the spinal dorsal horn after chronic morphine treatment for 7 days while D2DR antagonist sulpiride (8 μg/10 μl, i.t.) disrupted the interactions of the MOR/D2DR (n = 3).
Blockade of D2DR in spinal cord can disrupt the interactions between MOR and D2DR to attenuate morphine tolerance. These findings highlight the possibility of a new clinical strategy to prevent morphine antinociceptive tolerance.
Friday, August 28, 2015
Small Molecules/Peptides for Pain Researchers
The roots of Neuromics is providing solutions for the study of nociceptive and neuropathic pain. Our tools are widely used and frequently published. We are pleased to bring you these small molecules designed to help you better understand the roots of pain and potential therapies.
Name
|
Catalog #
|
Type
|
Size
|
Price
|
187-10
|
Agonist
|
10 mg
50 mg |
$165
$665 |
|
323-10
|
Antagonist
|
10 mg
50 mg |
$319
$1,319 |
|
1224-10
|
Modulator
|
10 mg
50 mg |
$99
$299 |
|
1062-10
|
Agonist
|
10 mg
|
$299
|
|
1116-10
|
Modulator
|
10 mg
50 mg |
$119
$449 |
|
3661-10
|
Antagonist
|
10 mg
50 mg |
$183
$795 |
|
699-10
|
Ligand
|
10 mg
1 mg |
$159
$609 |
|
1560-1
|
Antagonist
|
1 mg
|
$135
|
|
898-2
|
Antagonist
|
2 mg
|
$179
|
|
1055-5
|
Agonist
|
5 mg
|
$139
|
|
1056-5
|
Agonist
|
5 mg
|
$139
|
|
1480-10
|
Agonist
|
10 mg
10 mg |
$229
$929 |
|
0754-10
|
Agonist
|
10 mg
10 mg |
$199
$809 |
|
910-1
|
Inhibitor
|
1 mg
|
$179
|
|
1198-1
|
Inhibitor
|
1 mg
|
$195
|
Tuesday, July 21, 2015
Neuropeptides and Stress
Select Neuropeptides can help moderate stress and related Alcohol seeking behaviors. The authors of this study reference use of our ppENK and proDYN Neuropeptides: J.R. Schank,B.S. Nelson,R. Damadzic,J.D. Tapocik,M. Yao,C.E. King,K.E. Rowe,K. Cheng,K.C. Rice,M. Heili. Neurokinin-1 receptor antagonism attenuates neuronal activity triggered by stress-induced reinstatement of alcohol seeking. doi:10.1016/j.neuropharm.2015.07.009.
Image: Mouse striatum stained with D2 cell marker Enkephalin (RA14124) in green and with neuronal marker NeuN in red courtesy of Dr Heike Rebholz of City College of New York.
Our Pain/Inflammation Antibodies are widely used and frequently referenced in studies of stress and the intersection between stress and pain.
Tuesday, June 24, 2014
More Gains on Pain Research
There are multiple descriptions for chronic and acute pain: stabbing, burning, cutting, itching, numbing, crushing and more. Understanding the pathways are important to optimizing the therapies and treatments for the many different forms of pain.
The foundation of Neuromics is providing top notch pain research markers, gene expression analysis tools and cell based assays. Here I share recent publications from customers using these:
Inflammatory Joint Pain: Fiona B Carr, Sandrine M Géranton and Stephen P Hunt. Descending controls modulate inflammatory joint pain and regulate CXC chemokine and iNOS expression in the dorsal horn. Molecular Pain 2014, 10:39 doi:10.1186/1744-8069-10-39...mu opioid receptor, 1:10,000, Neuromics (RA10104)...
Temperature Sensation: Marics I, Malapert P, Reynders A, Gaillard S, Moqrich A (2014) Acute Heat-Evoked Temperature Sensation Is Impaired but Not Abolished in Mice Lacking TRPV1 and TRPV3 Channels. PLoS ONE 9(6): e99828. doi:10.1371/journal.pone.0099828. ...TRPV1 antibody (1/1000 dilution, Neuromics)...
Acute Pain: Paulino Barragán-Iglesias, Hector I. Rocha-González, Jorge Baruch Pineda-Farias, Janet Murbartián, Beatriz Godínez-Chaparro, Peter S. Reinach, Thiago M. Cunha, Fernando Q. Cunha, Vinicio Granados-Soto, Inhibition of peripheral anion exchanger 3 decreases formalin-induced pain, European Journal of Pharmacology, Available online 27 May 2014, ISSN 0014-2999, http://dx.doi.org/10.1016/j.ejphar.2014.05.029. (http://www.sciencedirect.com/science/article/pii/S0014299914003914). ...substance P (guinea pig; 1:200; Cat # GP14110; Neuromics, Edina, MN), and purinergic P2×3 receptor (guinea pig: 1:1000; Cat # GP10108 ...
We are pleased with the many publications and customer provided data proving the quality of our many pain research tools. I am always available to serve you, Pete Shuster (pshuster@neuromics.com) or direct phone: 612-801-1007.
Tuesday, January 28, 2014
Pot and Pain
The debate for many centers of "true medical benefits". That's why research on understanding analgesic pathways is so important. Ironically, this study was conducted by my friends at Université de Montréal and Université de Sherbrooke in Quebec Canada: J. Desroches, J.-F. Bouchard, L. Gendron, P. Beaulieu. Involvement of cannabinoid receptors in peripheral and spinal morphine analgesia ☆ Neuroscience, Volume 261, 7 March 2014, Pages 23–42. http://dx.doi.org/10.1016/j.neuroscience.2013.12.030.
These teams have proven expert is using our Opioid Receptor Antibodies in their pain research. Here's a synopsis:
•Analgesia is the most common feature shared by the cannabinoid and opioid systems.
•The role of the cannabinoid system in the morphine-induced analgesia is uncertain.
•Peripheral and intrathecal morphine analgesia is altered in cnr1KO and cnr2KO mice.
•This attenuation is neither caused by a MOP malfunction nor by its downregulation.
Images: Deletion of the CB1 or CB2 receptors has no effect on the expression of MOP in the spinal cord. Immunofluorescence of spinal MOP revealed that the expression of MOP in laminae I and II of the dorsal horn of the spinal cord did not differ between cnr1WT (A) and cnr1KO (B) mice or between cnr2WT (C) and cnr2KO mice (D).
Observations here further support the existence of interactions between the cannabinoid and opioid systems. The loss of peripheral and spinal morphine analgesia is apparently caused neither by a decrease in MOP spinal expression nor by altered binding properties or G protein coupling of this receptor in cnr1KO and cnr2KO mice. The mechanisms underlying the loss of morphine analgesia are not clear but could include the release of endogenous cannabinoids in structures along the pain pathway or a disrupted endocannabinoid tone.
It is important funding that enables researchers to understand the analgesic pathways of marijuana continues to grow. This research could yield better control of pain with reduced side effects.
Sunday, April 14, 2013
P2X3 Receptors and Migraine
Our P2X Receptor Markers continue to be used in interesting and novel ways. Here researchers use our P2X3 Receptor Antibody to study expression using primary rat ganlia cultures: Swathi K. Hullugundi,Michel D. Ferrari, Arn M. J. M. van den Maagdenberg, Andrea Nistri. Andrea Nistri. The Mechanism of Functional Up-Regulation of P2X3 Receptors of Trigeminal Sensory Neurons in a Genetic Mouse Model of Familial Hemiplegic Migraine Type 1 (FHM-1). PLoS ONE 8(4): e60677. doi:10.1371/journal.pone.0060677
Abstract: A knock-in (KI) mouse model of FHM-1 expressing the R192Q missense mutation of the Cacna1a gene coding for the α1 subunit of CaV2.1 channels shows, at the level of the trigeminal ganglion, selective functional up-regulation of ATP -gated P2X3 receptors of sensory neurons that convey nociceptive signals to the brainstem. Why P2X3 receptors are constitutively more responsive, however, remains unclear as their membrane expression and TRPV1 nociceptor activity are the same as in wildtype (WT) neurons. Using primary cultures of WT or KI trigeminal ganglia, we investigated whether soluble compounds that may contribute to initiating (or maintaining) migraine attacks, such as TNFα, CGRP, and BDNF, might be responsible for increasing P2X3 receptor responses. Exogenous application of TNFα potentiated P2X3 receptor-mediated currents of WT but not of KI neurons, most of which expressed both the P2X3 receptor and the TNFα receptor TNFR2. However, sustained TNFα neutralization failed to change WT or KI P2X3 receptor currents. This suggests that endogenous TNFα does not regulate P2X3 receptor responses. Nonetheless, on cultures made from both genotypes, exogenous TNFα enhanced TRPV1 receptor-mediated currents expressed by a few neurons, suggesting transient amplification of TRPV1 nociceptor responses. CGRP increased P2X3 receptor currents only in WT cultures, although prolonged CGRP receptor antagonism or BDNF neutralization reduced KI currents to WT levels. Our data suggest that, in KI trigeminal ganglion cultures, constitutive up-regulation of P2X3 receptors probably is already maximal and is apparently contributed by basal CGRP and BDNF levels, thereby rendering these neurons more responsive to extracellular ATP.
Images: Examples of TNFR2 and P2X3 co-exexpression in (wildtype) WT and R192Q (knockin) KI neurons. Left panel shows P2X3 expression (green), and right panel shows TNFR2 staining (red). B, Histograms quantifying % of cells co-expressing TNFR2 and P2X3: both WT and KI cultures show similar TNFR2 and P2X3 co-expression. N = 3 independent experiments (6 mice). C, Representative traces of currents induced by application of α,β-meATP (10 µM, 2 s) to WT or R192Q KI neurons in control conditions or after 4 h TNFα application. D, Histograms show average peak amplitudes of P2X3 receptor-mediated currents: WT control (open bar), n = 30; WT TNFα (stippled bar), n = 38; KI control (grey bar), n = 34; KI TNFα (stippled gray bar), n = 34; ** = p<0 .006="" i="" nbsp="" p="">doi:10.1371/journal.pone.0060677.g001.
Understanding the interplay between TNFR2 and P2X3 could lead to a better understanding of the root causes of migraines. This could open up yet more potential drug targets for this insidious condition.
Check out these related reagent categories:
All Purinergic Receptor Antibodies
Pain and Inflammation Research Antibodies
Neurotransmission Research Antibodies
Primary Neurons and Astrocytes-Primary human, rat and mouse neurons and astrocytes
Sunday, December 16, 2012
P2X Receptor Markers-Pubs Update
Here's a summary of publications: F.C. Pradoa, D. Araldia, A.S. Vieiraa, M.C.G. Oliveira-Fusarob, C.H. Tambelia, C.A. Parada. Neuronal P2X3 receptor activation is essential to the hyperalgesia induced by prostaglandins and sympathomimetic amines released during inflammation. http://dx.doi.org/10.1016/j.neuropharm.2012.11.011. non-fat dry milk at room temperature, followed by incubation with P2X3 or PKCɛ rabbit polyclonal antibody (1:500; Neuromics) overnight at 4 °C, rinsed six times with TBST, and then incubated for 40 min in goat anti-rabbit IgG peroxidase conjugate...
Min Liu, PhD, MD, Yun-fei Xu, PhD, MD, Yuan Feng, PhD, MD, Fengqiang Yang, MD, Jun Luo, MD, Wei Zhai, PhD, MD, Jian-ping Che, MD, Guang-chun Wang, MD, Jun-hua Zheng, PhD. Epigallocatechin gallate attenuates interstitial cystitis in human bladder urothelium cells by modulating purinergic receptors. Journal of Surgical Research. http://dx.doi.org/10.1016/j.jss.2012.11.041
...P2X2 (Neuromics, Northfield, MN)...
Abeer W Saeed, Alfredo Ribeiro-da-Silva. Non-peptidergic primary afferents are presynaptic to neurokinin-1 receptor immunoreactive lamina I projection neurons in rat spinal cord. Molecular Pain 2012, 8:64 doi:10.1186/1744-8069-8-64Anna M.W. Taylora, Maria Osikowicza, Alfredo Ribeiro-da-Silva. Consequences of the ablation of nonpeptidergic afferents in an animal model of trigeminal neuropathic pain. PAIN. Volume 153, Issue 6, June 2012, Pages 1311–1319. http://dx.doi.org/10.1016/j.pain.2012.03.023.
Anna M.W. Taylora, Maria Osikowicza, Alfredo Ribeiro-da-Silva. Consequences of the ablation of nonpeptidergic afferents in an animal model of trigeminal neuropathic pain. PAIN. Volume 153, Issue 6, June 2012, Pages 1311–1319. doi.org/10.1016/j.pain.2012.03.023...Sections were then incubated for 48 hours at 4°C with a guinea pig polyclonal anti-P2X3 antibody (1:25,000; Neuromics, Edina, MN, USA), diluted in PBS-T. Following primary antibody incubation, sections were treated with a biotin-conjugated...
Ji Z-G , Ito S , Honjoh T , Ohta H , Ishizuka T , et al. 2012 Light-evoked Somatosensory Perception of Transgenic Rats That Express Channelrhodopsin-2 in Dorsal Root Ganglion Cells. PLoS ONE 7(3): e32699...guinea-pig anti-P2X3 (1:1,000, GP10108, Neuromics, Edina, MN, USA)...
Sunday, October 21, 2012
P2X3 Receptor and CGRP Antibodies Immunostaining
Here, use of our rabbit anti-CGRP and guinea pig anti-P2X3 is referenced. Please note the high titer of these antibodies (dilution is 1:25,000): Abeer W Saeed, Alfredo Ribeiro-da-Silva. Non-peptidergic primary afferents are presynaptic to neurokinin-1 receptor immunoreactive lamina I projection neurons in rat spinal cord. Molecular Pain 2012, 8:64 doi:10.1186/1744-8069-8-64.
Images: CGRP, IB4 and P2X3 staining in transverse spinal cord sections. A and B show low magnification confocal images of CGRP-IR and IB4 positive (A) or P2X3-IR (B) fibers. C and D represent high magnification confocal images from the middle third of the lateromedial extent of the superficial dorsal horn. In C, note that there is limited co-localization of IB4 and CGRP (in yellow). Arrowheads show axonal varicosities (boutons) from nonpeptidergic fibers in lamina I, which do not co-localize CGRP immunoreactivity. The framed regions in A and B indicate the approximate regions from where C and D, respectively, were obtained (the latter originate from other sections). CGRP (in green); IB4 (in red); P2X3 (in red). Scale bar (A, B) = 200 μm; scale bar (C, D) = 20 μm
Tissue processing: The injection site at the level of the parabrachial nucleus was examined by cutting serial, 100 μm-thick coronal sections of the relevant brain region. The dorsal aspect of the L4-L5 spinal cord segment was cut into serial, 50 μm-thick horizontal sections (n = 10), 50 μm-thick parasagittal sections (n = 4) or 50 μm-thick transverse sections (n = 4). All sections were cut using a freezing sledge microtome (Leica, Richmond Hill, Ontario) and collected as freefloating in phosphate-buffered saline (PBS) with 0.2% Triton-X 100 (PBS + T). To block unspecific staining, all spinal cord sections were incubated, for one hour, in 10% normal donkey serum (NDS) (Jackson, West Grove, PA) in PBS + T at room temperature. Subsequently, the sections were placed in primary antibodies (or conjugated lectin IB4 - see below) for 48 hours at 4 °C. We used a mixture of 2 or 4 primary antibodies (each raised in a different species), or IB4, in PBS + T containing 5% NDS. Next, the sections were washed in PBS + T and then incubated in species-specific secondary antibodies that were raised in donkey and conjugated to either AlexaFluor 488, AlexaFluor 405, Rhodamine RedX or biotin. The sections were incubated in 3 different cocktails: #1) rabbit anti-CGRP at a 1:200 dilution (Sigma, St Louis, MO) and lectin IB4 conjugated to AlexaFluor 568 at a 1:200 dilution (Molecular Probes); #2) rabbit anti-CGRP and guinea pig anti-P2X3 at a 1:25,000 dilution (Neuromics, Edina, MN); #3) goat anti-CTb at a 1:5000 dilution (List Biological), rabbit anti-NK-1r at a 1:10000 dilution (Sigma, St Louis, MO), guinea pig anti-CGRP at a 1:8000 dilution (Peninsula, San Carlos, CA) and lectin IB4 conjugated to AlexaFluor 647 at a 1:200 dilution (Molecular Probes). All the sections were washed with PBS + T and then (for #1) incubated for 2 hours at room temperature with donkey anti-rabbit AlexaFluor 488; (for #2) incubated for 90 minutes in a biotin conjugated donkey anti-guinea pig IgG (Jackson Immunoresearch, West Grove, PA, 1:200). Further signal amplification was achieved by treating the sections with 1 hour incubation in an avidin-biotin (A + B) complex (Vectastain Elite ABC kit, Vector Laboratories) followed by tyramide (Perkin-Elmer, Norwalk, CT, 1:75) for 7 minutes. Sections were then incubated in streptavidin conjugated to AlexaFluor 568 (Molecular Probes, Eugene, OR, 1:200) and donkey anti-rabbit AlexaFluor 488; or (for #3) incubated for 2 hours at room temperature with secondary antibodies: donkey anti-goat Rhodamine Red X, donkey anti-rabbit AlexaFluor 488, and donkey anti-guinea pig AlexaFluor 405. Finally, sections were washed with PBS, mounted on gelatin-subbed slides and coverslipped with an anti-fading mounting medium (Aqua Polymount; Polysciences, Warrington, PA). Slides were stored at −4 °C pending further processing.
I will continue to publish outstanding customer data/images using our natibodies/markers.
Wednesday, June 20, 2012
TRPA1 and Tooth Pain
Transient receptor potential ankyrin 1 (TRPA1) is activated by noxious cold (<17°C) and contributes to cold and mechanical hypersensitivity after inflammation and nerve injury: Yun Sook Kim, PhD, Hoon Kap Jung, DDS, Tae Kyung Kwon, DDS, Chin Soo Kim, DDS, PhD, Jin Hyun Cho, DDS, PhD, Dong Kuk Ahn, DDS, PhD, Yong Chul Bae, DDS, PhD.Expression of Transient Receptor Potential Ankyrin in Human Dental Pulp. Journal of Endodontics. Available online 8 June 2012. doi.org/10.1016/j.joen.2012.04.024.
Highlights: TRPA1 was expressed in a large number of axons branching extensively in the peripheral pulp and in a few axons within the nerve bundles in the core of the coronal pulp and in the radicular pulp. Under electron microscopy, TRPA1 immunoreactivity was typically localized near the plasma membrane of unmyelinated axons in the peripheral pulp, suggesting that in these axons it may act as a functional receptor. The proportion of axons expressing TRPA1 in neurofilament 200–positive axons significantly increased in the painful pulp compared with the normal pulp. TRPA1 was also densely expressed in the processes and the cell body of odontoblasts. A large number of axons coexpressed TRPA1 and Nav1.8.
Pain and Inflammation Research Antibodies
Neurotransmission -Neurotransmission Research Antibody Categories
Wednesday, July 06, 2011
Guinea Pig P2X3 Update-Good News
We tried and tried to re-make it. The result was none of the bleeds we tested had a signal strong enough to release the antibody. We had a customer suggest re-testing several of the more promising bleeds. Thank you! We have good news on results and we are offering for 50% off. This is to acknowledge the investment required for TSA and Guinea Pig Biotinylated Antibody.
Here're the recent pubs I referenced:
Gabriela Castañeda-Corral, Héctor I. Rocha-González, Beatriz Godínez-Chaparro, Juan Miguel Jiménez-Andrade and Vinicio Granados-Soto. Role of the spinal Na+/H+ exchanger in formalin-induced nociception. Neuroscience Letters. doi:10.1016/j.neulet.2011.06.048....SP (guinea pig; 1:500; Cat# GP14110; Neuromics), CGRP (goat, 1:500; Cat# Ab36001; Abcam) and P2X3 receptor (guinea pig: 1:10,000; Cat# GP10108; Neuromics)...
Anna M.W. Taylora and Alfredo Ribeiro-da-Silva. GDNF levels in the lower lip skin in a rat model of trigeminal neuropathic pain: Implications for nonpeptidergic fiber reinnervation and parasympathetic sprouting. PAIN Volume 152, Issue 7, July 2011, Pages 1502-1510. doi:10.1016/j.pain.2011.02.035.
...Sections were then incubated for 48h at 4°C with a guinea pig polyclonal anti-P2X3 (1:25,000; Neuromics, Edina, MN)...
Thursday, November 18, 2010
Cancer-Induced Bone Pain
Image: Image:Shows rat mixed neuron/glial cultures stained with mouse monoclonal antibody to neurofilament subunit NF-L clone 7D1 (green) and chicken antibody to neurofilament NF-H. This antibody binds primarily to the phosphorylated axonal forms of NF-H, in contrast to the NF-L antibody which stains both axonal and dendritic/perikaryal neurofilaments. The NF-L antibody therefore reveals a prominent cell body in green, while the surrounding axonal profiles are orange, since the are bound by both NF-L and the chicken NF-H antibody. Blue is a DNA stain. Protocol on data sheet.
Juan M. Jimenez-Andrade, Aaron P. Bloom, James I. Stake, William G. Mantyh, Reid N. Taylor, Katie T. Freeman, Joseph R. Ghilardi, Michael A. Kuskowski, and Patrick W. Mantyh Pathological Sprouting of Adult Nociceptors in Chronic Prostate Cancer-Induced Bone Pain. J. Neurosci., Nov 2010; 30: 14649 - 14656 ; doi:10.1523/JNEUROSCI.3300-10.2010
Here're several other pubs referencing use of our antibodies in studying bone cancer pain:
Kyle G. Halvorson, BA, Molly A. Sevcik, BA, Joseph R. Ghilardi, BS, BA, Lucy J. Sullivan, BA, Nathan J. Koewler, BS, Frieder Bauss, PhD, and Patrick W. Mantyh, PhD. Intravenous Ibandronate Rapidly Reduces Pain, Neurochemical Indices of Central Sensitization, Tumor Burden, and Skeletal Destruction in a Mouse Model of Bone Cancer. Published online 2008 April 14. doi: 10.1016/j.jpainsymman.2007.10.005
...pro-dynorphin (DYN, polyclonal guinea pig anti-rat, 1:1,000; Neuromics, Minneapolis, MN)...
Timothy K. Y. Kaan, Ping K. Yip, Sital Patel, Meirion Davies, Fabien Marchand, Debra A. Cockayne, Philip A. Nunn, Anthony H. Dickenson, Anthony P. D. W. Ford, Yu Zhong, Marzia Malcangio, and Stephen B. McMahon Systemic blockade of P2X3 and P2X2/3 receptors attenuates bone cancer pain behaviour in rats. Brain, September 2010; 133: 2549 - 2564.
......Slides were then incubated with rabbit anti-P2X3 (1:2000, Neuromics) and sheep anti-calcitonin gene-related peptide (1:1000, Biomol...anti-beta-III-tubulin (1:4000, Promega) and guinea pig anti-P2X3 (1:100, Neuromics). The next day, after three washes with phosphate-buffered......
I will keep you posted on this important topic.
Thursday, July 29, 2010
Let-7 microRNAs and Nociceptive Pain
We want to recognize Dr. Zaijie Jim Wang and his team for being the first to use our Mu Opioid Receptor for studying the potential role of microRNAs in Nociception.
Ying He, Cheng Yang, Chelsea M. Kirkmire, and Zaijie Jim Wang. Regulation of Opioid Tolerance by let-7 Family MicroRNA Targeting the µ Opioid Receptor. The Journal of Neuroscience, July 28, 2010, 30(30):10251-10258; doi:10.1523/JNEUROSCI.2419-10.2010
Abstract: MicroRNA has emerged as a critical regulator of neuronal functions. This study aimed to test whether let-7 microRNAs can regulate the µ opioid receptor (MOR) and opioid tolerance. Employing bioinformatics, we identified a let-7 binding site in the 3'-untranslated region (UTR) of MOR mRNA, which was experimentally confirmed as a direct target of let-7. The repressive regulation of MOR by let-7 was revealed using a LNA-let-7 inhibitor to knockdown let-7 in SH-SY5Y cells. Conversely, morphine significantly upregulated let-7 expression in SH-SY5Y cells and in a mouse model of opioid tolerance. The LNA-let-7 inhibitor decreased brain let-7 levels and partially attenuated opioid antinociceptive tolerance in mice. Although chronic morphine treatment did not change overall MOR transcript, polysome-associated mRNA declined in a let-7-dependent manner. let-7 was identified as a mediator translocating and sequestering MOR mRNA to P-bodies, leading to translation repression. These results suggest that let-7 plays an integral role in opioid tolerance.
- Western blot analysis. Western blot analysis was performed as previously described (Tang et al., 2006) using the anti-µ opioid receptor antibody (1:1000; Neuromics). The expression of β-actin was similarly determined from the same blots using a monoclonal antibody (1:10,000; Sigma).
- For immunofluorescence analysis, the antibody for hDcp1a (Santa Cruz Biotechnology) and MOR were used at 1:500 and 1:5000 dilutions, respectively. Secondary anti-goat and anti-mouse antibodies labeled with Alexa 488 and Alexa 594 fluorochromes (Invitrogen), respectively, were used at 1:500 dilutions.
All Opioid Receptor Antibodies
Pain and Inflammation
Neurotransmission Research Antibodies-GPCRs, Ligand Gated Ion Channels,
Biogenic Amines and more
i-Fect Transfection Kit -gene silencing of DOR,
NaV1.8 tetrodotoxin-resistant sodium channel, NTS2 and more in-vitro and in vivo
Primary Neurons and Astrocytes-Primary human,
rat and mouse neurons and astrocytes
Monday, July 26, 2010
δ- and μ-opioid receptors co-expression and Nociceptive Pain
Hai-Bo Wanga, Bo Zhaoa, Yan-Qing Zhonga, Kai-Cheng Li, Zi-Yan Li, Qiong Wang, Yin-Jing Lua, Zhen-Ning Zhang, Shao-Qiu He, Han-Cheng Zheng, Sheng-Xi Wu, Tomas G. M. Hökfelt, Lan Baob, and Xu Zhanga. Coexpression of δ- and μ-opioid receptors in nociceptive sensory neurons. PNAS July 20, 2010 vol. 107 no. 29 13117-13122.
Immunostaining. Adult rats, mice, and Oprd1 exon 1-deleted mice were fixed. Cryostat sections of L4 and L5 DRGs and spinal cord segments were processed for immunofluorescence staining (13) with Rb anti-DOR13–17 (1:2,000–1:60,000; DiaSorin and 1:4,000–1:60,000; Neuromics), Rb anti-DOR12–18 (1:30,000–1:120,000; Alomone), Rb anti-DOR1358–372 (1:1,000–1:2,000; Lifespan Biosciences), Rb anti-MOR (1:1,000; Neuromics); guinea pig anti-SP (1:500; Neuromics), and mouse anti-CGRP (1:1,000; Biogenesis) antibodies. IB4-labeling was carried out with fluorescein-labeled GSL I-IB4 (1:200). The Myc-DOR1–transfected HEK293 cells and neurons were fixed and processed with mouse anti-Myc antibodies (1:500; DSHB). Nuclear DAPI staining was used to indicate HEK293 cells in control experiments.
Images: Distinct distribution patterns of DORs in subsets of DRG neurons of mice. Immunostaining with antibodies against DOR13–17 [A: 1:30,000, antibody 1 (ab #1); DiaSorin and C: antibody 2 (ab #2); Neuromics] shows DORs in small DRG neurons and afferent fibers in spinal laminae I–II. This immunostaining pattern is abolished by the antiserum preabsorption or the deletion of Oprd1 exon 1. Reduction in immunostaining is quantitatively assayed by determining the percentage of positive DRG neurons (B; n = 6) and fluorescence intensity (Ifluo.) in the laminae I–II (D; n = 5). **P < 0.01; ***P < 0.001. (Scale bars: A and C, 40 μm.). DOR labeling (anti-DOR13–17, 1:30,000; DiaSorin) associated with vesicles in peptidergic small DRG neurons (E and F) is absent in Oprd1 exon 1-deleted mice (G). Colocalization of DORs and neuropeptides is shown by correlated peaks of Ifluo. measured along lines. (Scale bar: 8 μm.) (H) Immunostaining with antibodies against DOR12–18 (1:60,000; Alomone) shows the presence of DORs on the cell surface of large DRG neurons of mice. (Scale bar: 25 μm.) This staining pattern is abolished by preabsorption and is absent in Oprd1 exon 1-deleted mice. (Scale bar: 80 μm.) (I) Triple-immunostaining shows that DOR+ large DRG neurons contain neither SP nor CGRP. (Scale bar: 80 μm.)
Immunoblotting.The samples were processed for SDS/PAGE, transferred, probed with Rb antibodies against MOR (1:500; Neuromics), phospho-DOR1 (1:1,000; Neuromics), phospho-MOR (1:1,000; Neuromics), Myc (1:500; DSHB), Flag (1:1,000; Sigma), or actin (1:50,000; Chemicon) and visualized with enhanced chemiluminescence (19).
| Featured and Related Reagents: Mu Opioid Receptor-Rabbit Mu Opioid Receptor-Guinea Pig Delta Opioid Receptor 3-17 Delta Opioid Receptor 358-372 Delta Opioid Receptor 358-372 Kappa Opioid Receptor phospho-Mu Opioid Receptor (Ser375) MOR-1C ORL 1-Pure | ORL1-Whole Serum All Opioid Receptor Antibodies Pain and Inflammation Neurotransmission Research Antibodies-GPCRs, Ligand Gated Ion Channels, Biogenic Amines and more i-Fect Transfection Kit -gene silencing of DOR, NaV1.8 tetrodotoxin-resistant sodium channel, NTS2 and more in-vitro and in vivo Primary Neurons and Astrocytes-Primary human, rat and mouse neurons and astrocytes |
Friday, February 19, 2010
Making Gains on Pain Research
Hua Zhang and A. S. Verkman. Aquaporin-1 Tunes Pain Perception by Interaction with Nav1.8 Na+ Channels in Dorsal Root Ganglion Neurons. February 19, 2010 The Journal of Biological Chemistry, 285, 5896-5906.
...chicken anti-calcitonin gene-related peptide (CGRP; 1:500, Neuromics, Edina, MN)...
Nathaniel A. Sowa, Bonnie Taylor-Blake, and Mark J. Zylka. Ecto-5'-Nucleotidase (CD73)  Inhibits Nociception by Hydrolyzing AMP to Adenosine in Nociceptive Circuits. The Journal of Neuroscience, February 10, 2010, 30(6):2235-2244; doi:10.1523/JNEUROSCI.5324-09.2010.
Inhibits Nociception by Hydrolyzing AMP to Adenosine in Nociceptive Circuits. The Journal of Neuroscience, February 10, 2010, 30(6):2235-2244; doi:10.1523/JNEUROSCI.5324-09.2010.
...rabbit anti-P2X3-RA10109, Neuromics; 1:750), rabbit anti- VR1 C-Terminus (TRPV1) - mouse specific (RA14113, Neuromics; 1:750
Images: Images: Confocal images showing the effect of RTX on mu opioid receptor and TRPV1 immunoreactive DRG neurons and afferent terminals in the spinal cord. A: representative confocal images showing mu opioid receptor (green) and TRPV1 (red) immunoreactivities in DRG neurons of one vehicle- and one RTX-treated rat. Scale bar, 40 um. B: confocal images showing mu opioid receptor (green) and TRPV1 (red) immunoreactivities in afferent terminals in the spinal dorsal horn of 1 vehicle- and 1 RTX-treated rat. Scale bar, 80 um. Inset: high-magnification images (scale bar = 5 um) showing co-localization of mu opioid receptor and TRPV1 immunoreactivity in the lamina I. Co-localization of the mu opioid receptor and TRPV1 immunoreactivity is indicated in yellow when 2 images are digitally merged. All images are single confocal optical sections. Shao-Rui Chen and Hui-Lin Pan. Loss of TRPV1-Expressing Sensory Neurons Reduces Spinal mu-Opioid Receptors But Paradoxically Potentiates Opioid Analgesia. doi:10.1152/jn.01343.2005.
Neuromics' i-Fect ™ siRNA Transfection Reagent continues to be used a tool for studying expression of genes suspected to paly a role in pain . Expression studies include: DOR , The β3 subunit of the Na+,K+-ATPase, rSNSR1, NTS1. NAV1.8, CaV1.2 and more.
Here's a link to all transfection publications: Transfection Kit Pubs
Neurotransmission Research Antibodies-GPCRs, Ligand Gated Ion Channels, Biogenic Amines and more | Purinergic Receptors Primary Neurons and Astrocytes- Primary human, rat and mouse neurons and astrocytes by Category |
Saturday, October 17, 2009
More Success with TRPV1
The antibody was used for both Western Blot and Immunohistochemistry. Here's a recent publication of their results:
Vanessa Bockhart, Cristina Elena Constantin, Annett Häussler, Nina Wijnvoord, Maike Kanngiesser, Thekla Myrczek, Geethanjali Pickert,Laura Popp, Jürgen-Markus Sobotzik, Manolis Pasparakis, Rohini Kuner, Gerd Geisslinger, Christian Schultz, Michaela Kress, and Irmgard Tegeder. Inhibitor B Kinase β Deficiency in Primary Nociceptive Neurons Increases TRP Channel Sensitivity. The Journal of Neuroscience, October 14, 2009, 29(41):12919-12929; doi:10.1523/JNEUROSCI.1496-09.2009
Related Reagents:
All TRPV (Vanilloid); TRPM; TRPA and TRPC Antibodies
Pain and Inflammation Research Antibodies
Monday, September 28, 2009
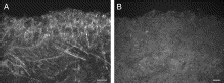
Prodynorphin at Work

Image: Staining of adult rat spinal cord.
The tissue is perfusion-fixed (4% freshly prepared formaldehyde) adult rat spinal cord, reacted overnight with the PPD at 1:1000 and then o/n in Alexa488 secondary (raised in donkey, Invitrogen, 1:500).
The confocal image stack was taken through a 60x oil lens (Bio-Rad Radiance confocal) - pixel size is 0.196 micrometre and this is a projection of 10 confocal optical sections at 0.5 micrometre z-spacing.
Customer Publications
Related Reagents:
proDynorphin (guinea pig)
Opioid Receptors
Pain and Inflammation Antibodies
Labels
Protocols Online
Alzforum News
About Me
Tracker
ShareThis
Followers
My Blog List
-
Scientists grow retina cells from skin-derived stem cells - WASHINGTON - University of Wisconsin-Madison researchers have successfully grown multiple types of retina cells from two types of stem cells, giving new ho...16 years ago
-
Should Pfizer buy Wyeth? - Pfizer has reached the top of the pharmaceutical ladder in large part thanks to mega-acquisitions. The Warner Lambert acquisition most notably brought Lipi...17 years ago














