P2X3 Activates TRPA1, 5-HT3 and 5-HT1A Receptors
Endogenous ATP via activation of
P2X3 Receptors contributes to inflammatory nociception in different models, including the formalin injected in subcutaneous tissue of the rat's hind paw. In this study, researchers evaluated whether TRPA1, 5-HT3 and 5-HT1A receptors, whose activation is essential to formalin-induced inflammatory nociception, are involved in the nociception induced by activation of P2X3 receptors on subcutaneous tissue of the rat's hind paw:
Suzy Krimon, Dionéia Araldi, Filipe César do Prado, Cláudia Herrera Tambeli, Maria Cláudia G. Oliveira-Fusaro, Carlos Amílcar Parada. P2X3 receptors induced inflammatory nociception modulated by TRPA1, 5-HT3 and 5-HT1A receptors. Pharmacology Biochemistry and Behavior, Available online 8 October 2013. http://dx.doi.org/10.1016/j.pbb.2013.09.017. Our widely used and frequently published P2X3 R Antibody places a central role in measuring the expression of the protein...containing 5% non-fat dry milk at room temperature, followed by incubation with P2X3 rabbit polyclonal antibody (1:500; Neuromics) overnight at 4 °C, rinsed six times with TBST, and then incubated for 40 minutes in goat anti-rabbit IgG peroxidase...
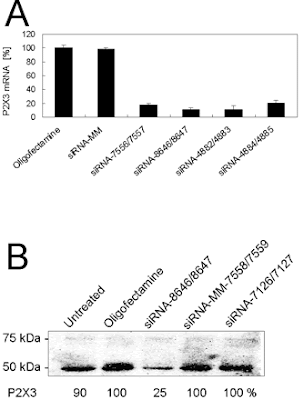
Image: Neuromics' P2X3 R WB Example: Sequence‐specific siRNA‐mediated repression of P2X3. (A) P2X3 mRNA inhibition by 200 nM siRNA duplexes. Twenty‐four hours after transfection of CHO‐rP2X3 cells, P2X3‐specific mRNA was measured with Q‐PCR and plotted as percentage of mRNA detected in the control treated with Oligofectamine alone. Sequences and modifications are shown in Figure 1B and Table 1. (B) P2X3 protein reduction by 200 nM siRNA‐8646/8647, but not by its mismatch analogue siRNA‐MM‐7558/7559 or the unrelated siRNA‐7126/7127. Twenty‐four hours after transfection, protein was extracted and analysed by western blotting. P2X3‐specific immunodetection reveals expression levels as shown below (an average value from two experiments). Time points as indicated at the top. Molecular weights of two glycosylated forms of P2X3 are shown on the left.
Conclusions: Nociceptive response intensity was measured by observing the rat's behavior and considering the number of times the animal reflexively raised its hind paw (flinches) in 60 min. Local subcutaneous administration of the selective TRPA1, 5-HT3 or 5-HT1A receptor antagonists HC 030031, tropisetron and WAY 100,135, respectively, prevented the nociceptive responses induced by the administration in the same site of the non-selective P2X3 receptor agonist αβmeATP. Administration of the selective P2X3 and P2X2/3 receptor antagonist A-317491 or pretreatment with oligonucleotides antisense against P2X3 receptor prevented the formalin-induced behavioral nociceptive responses during the first and second phases. Also, the co-administration of a subthreshold dose of αβmeATP with a subthreshold dose of formalin induced nociceptive behavior, which was prevented by local administration of tropisetron, HC 030031 or WAY 100, 135. These findings have demonstrated that the activation of P2X3 receptors induces inflammatory nociception modulated by TRPA1, 5-HT3 and 5-HT1A receptors. Also, they suggest that inflammatory nociception is modulated by the release of endogenous ATP and P2X3 receptor activation, which in turn, increases primary afferent nociceptor susceptibility to the action of inflammatory mediators via interaction with TRPA1, 5-HT3 and 5-HT1A receptors in the peripheral tissue.
I will continue to post
pain and inflammation related studies that reference the use of our
antibodies.






