We want to recognize Dr. Zaijie Jim Wang and his team for being the first to use our Mu Opioid Receptor for studying the potential role of microRNAs in Nociception.
Ying He, Cheng Yang, Chelsea M. Kirkmire, and Zaijie Jim Wang. Regulation of Opioid Tolerance by let-7 Family MicroRNA Targeting the µ Opioid Receptor. The Journal of Neuroscience, July 28, 2010, 30(30):10251-10258; doi:10.1523/JNEUROSCI.2419-10.2010
Abstract: MicroRNA has emerged as a critical regulator of neuronal functions. This study aimed to test whether let-7 microRNAs can regulate the µ opioid receptor (MOR) and opioid tolerance. Employing bioinformatics, we identified a let-7 binding site in the 3'-untranslated region (UTR) of MOR mRNA, which was experimentally confirmed as a direct target of let-7. The repressive regulation of MOR by let-7 was revealed using a LNA-let-7 inhibitor to knockdown let-7 in SH-SY5Y cells. Conversely, morphine significantly upregulated let-7 expression in SH-SY5Y cells and in a mouse model of opioid tolerance. The LNA-let-7 inhibitor decreased brain let-7 levels and partially attenuated opioid antinociceptive tolerance in mice. Although chronic morphine treatment did not change overall MOR transcript, polysome-associated mRNA declined in a let-7-dependent manner. let-7 was identified as a mediator translocating and sequestering MOR mRNA to P-bodies, leading to translation repression. These results suggest that let-7 plays an integral role in opioid tolerance.
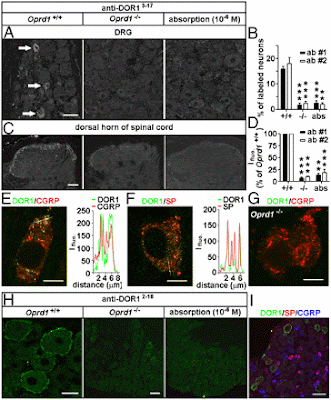
- Western blot analysis. Western blot analysis was performed as previously described (Tang et al., 2006) using the anti-µ opioid receptor antibody (1:1000; Neuromics). The expression of β-actin was similarly determined from the same blots using a monoclonal antibody (1:10,000; Sigma).
- For immunofluorescence analysis, the antibody for hDcp1a (Santa Cruz Biotechnology) and MOR were used at 1:500 and 1:5000 dilutions, respectively. Secondary anti-goat and anti-mouse antibodies labeled with Alexa 488 and Alexa 594 fluorochromes (Invitrogen), respectively, were used at 1:500 dilutions.
Related Reagent Links:
All Opioid Receptor Antibodies
Pain and Inflammation
Neurotransmission Research Antibodies-GPCRs, Ligand Gated Ion Channels,
Biogenic Amines and more
i-Fect Transfection Kit -gene silencing of DOR,
NaV1.8 tetrodotoxin-resistant sodium channel, NTS2 and more in-vitro and in vivo
Primary Neurons and Astrocytes-Primary human,
rat and mouse neurons and astrocytes
All Opioid Receptor Antibodies
Pain and Inflammation
Neurotransmission Research Antibodies-GPCRs, Ligand Gated Ion Channels,
Biogenic Amines and more
i-Fect Transfection Kit -gene silencing of DOR,
NaV1.8 tetrodotoxin-resistant sodium channel, NTS2 and more in-vitro and in vivo
Primary Neurons and Astrocytes-Primary human,
rat and mouse neurons and astrocytes